Here is how to train your vascular system to stay fully functional for the rest of your life.
Table of content:
- Why you can’t generalize study results to you, the individual
- Why the lack of generalizability is a good thing
- How to train for health, functionality, and independence
Introduction
There is this belief that the insights of clinical trials can be translated to individual people. It is as mistaken as it is widespread [1,2]. And it has two disastrous consequences.
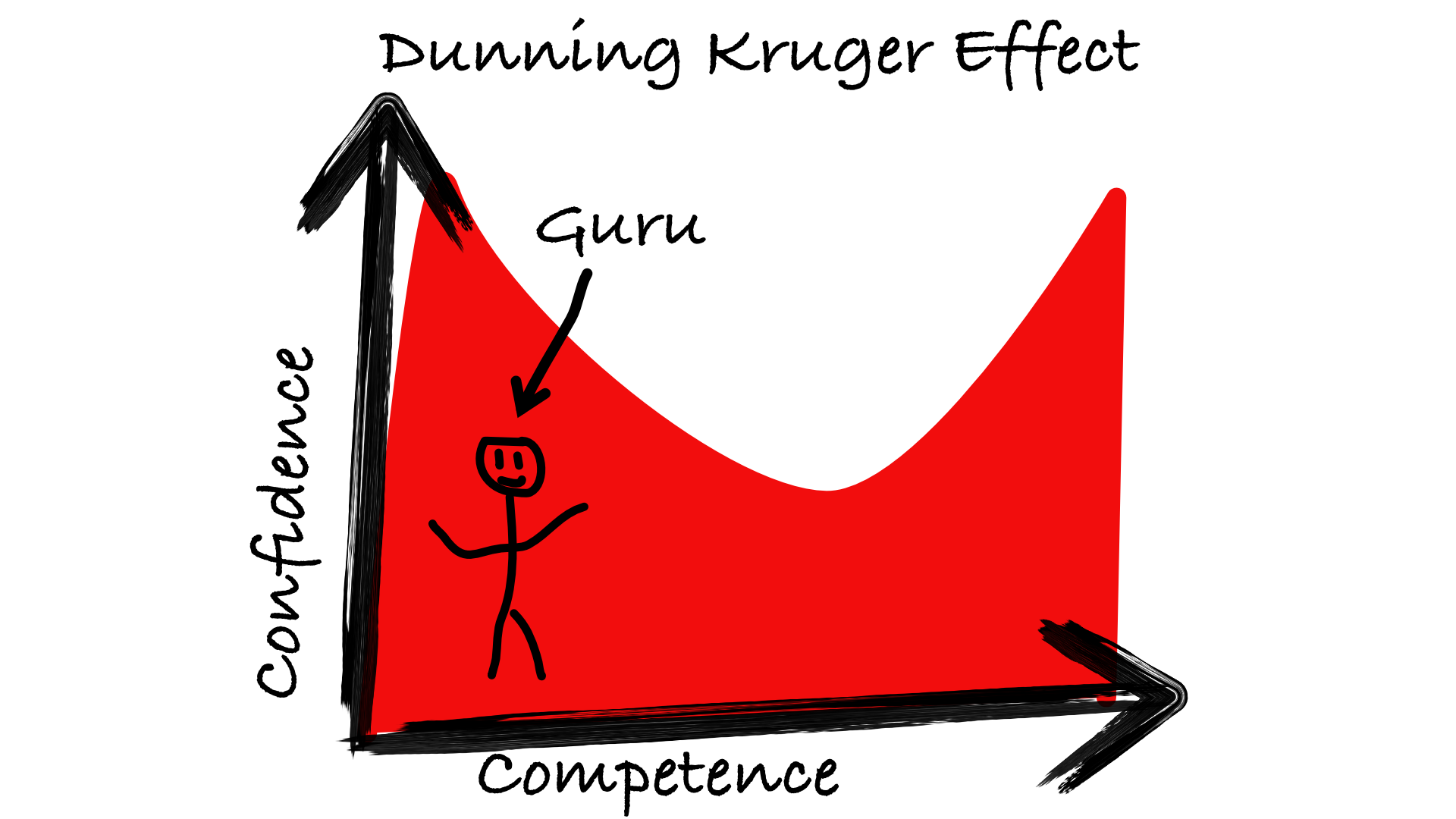
- The emergence of a caste of self-proclaimed health gurus too incompetent to spot the flaws in their renderings of medical study results.
- Most people who follow the incompetents’ advice fail to achieve the health benefits that they could otherwise realize if given accurate guidance.
The first is just a classic Dunning-Kruger effect: high confidence because of too little knowledge to spot one’s own incompetence.
“Ignorance more frequently begets confidence than does knowledge” Charles Darwin
The second is particularly worrying when the intended health benefit is to escape the ailments of aging: heart disease, stroke, diabetes, dementia, frailty, and cancer.
Most of us could avoid all these through ideal lifestyle choices. But what is ideal for me, will possibly not work for you.
Since life does not come with a reset button, we need to know what works and what doesn’t BEFORE we reach the point of no return.
In the following, I will address:
- why generalizing study results to you, the individual, is fatally flawed
- why this lack of generalizability is a good thing
- how you can realize the envisioned health benefits with surgical precision
Why you can’t generalize study results to you, the individual
If you have been exercising regularly for many years, you’ll probably have noticed it: the sensation that you have somehow slowed down your biological clock.
Science says this is more than just a sensation.
Researchers compared the endothelial cells (EC) of physically active 60-year-olds with those of
- healthy but sedentary 22-year-olds
- healthy but sedentary same-age peers
Explanatory Note 1
What is so special about the endothelial cells?
The Endothelium is the innermost lining of our blood vessels (arteries and veins, summarily called the vasculature). Its integrity makes the difference between protecting us from cardiovascular disease (CVD) and promoting it. Since CVD is heavily associated with aging it makes good sense to look at the endothelial cells (EC).
While the ECs of the physically active 60-year-olds were indistinguishable from those of the 22-year-olds, the cells of the sedentary 60-year-olds were biologically much older than those of both other groups [3].
The authors:
“The results … provide novel evidence in humans for increased endothelial cell (EC) senescence with healthy aging and a lack of increase in older adults who perform habitual aerobic exercise.”
In other words, people who exercise have endothelial cells that appear to be as young as those of sedentary people who are 40 years younger. No such luck for the sedentary older ones. Or at least, so it seems.
The study authors illustrated their findings with graphs like the one below.
Figure 1:
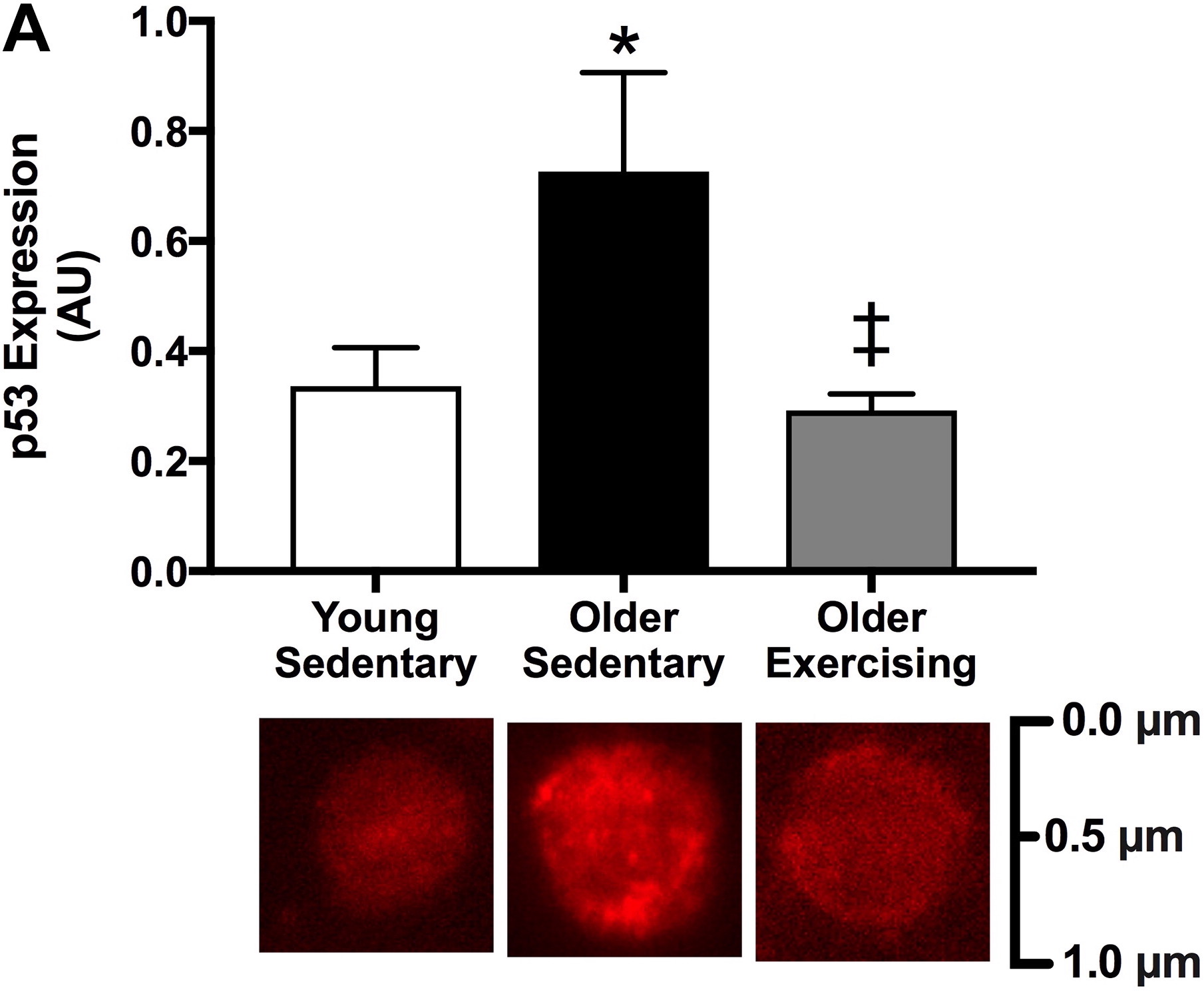
The older sedentary group is impressively different from the other two.
That’s where the key messages come from:
- “no difference between old trained and young sedentary groups”
- “big difference between old sedentary and old trained groups”
These statements are perfectly correct because the researchers compare groups of people who differ on defined characteristics (in this case age and exercise history).
Here is what charts like these conceal:
Differences in the mean separate groups from each other, not individuals.
The problem with statistics
Let’s visualize the data in a slightly different way (see the chart below). I used the authors’ published data to simulate the three study groups (it’s a legitimate statistical exercise of drawing sample populations from known data).
Figure 2
Each dot represents the p53 value of a participant. I have separated them into the three groups. The chart’s most important message is the substantial overlap between the groups.
Five of the 14 members (36%) of the old-trained group have values within the same range as the old-sedentary group (the last dot represents two people with nearly identical values).
All the old-sedentary participants lie within the range of the old-trained group, with 72% of them being well within the range of the young-sedentary group.
That makes the average of biomarkers such as p53 useless to guide your personal lifestyle habits. Or to judge their effectiveness.
Because, while there is an average, the average participant does not exist. Neither in the research setting nor “in the wild” so to speak.
The graph below illustrates my point.
Figure 3
Only participant No. 2 represents the average, just by chance. Participants 1 and 12 are close, but all the others are way off. That’s a picture you’ll see in every single medical study. It is the reason why you can’t generalize the results of group-based studies to you, the individual [1,2].
I promised to tell you…
Why the lack of generalizability is a good thing
Look at figure 2 again.
Now let’s assume that p53 really somehow reflects biological age (which it doesn’t, and I’ll explain why, but let’s not get distracted now).
At age 60+, with a value of 0.5, you either have trained not very effectively (if you belong to the trained group), or very efficiently if you belong to the “sedentary” group.
Here is why:
To be considered “trained”, participants had to have a history of
“…vigorous aerobic exercise including competitive cycling, running, and/or triathlons, for ≥5 days/wk and ≥45 min/session for at least the preceding 5 years”.
That’s a tall order.
But the sedentary-old were physically more active than the “sedentary” label implies: the condition was: aerobic exercise yes, but not more than twice weekly and not more than altogether 60 minutes per week. And there was no limitation on strength training.
As you can see from the chart, people with vastly different training histories can end up with the same p53 biological age indicator of, say, 0.5.
To the exercise extremist, that’s a meager dividend for an enormous effort. An effort that has rewarded his or her peers with a much better score. In comparison, the far less active “sedentary” person essentially sleep-walks to the same 0.5 mark.
Unfair, isn’t it?
No!
It’s not about fairness; it’s about mismatch.
If you and I can have the same result with two different exercise habits, and we can have different results with the same habit, then it’s not the habit alone that determines our individual results. Rather, it is how well each habit matches our individual predisposition. That should encourage you to find your personal best match. It will neither waste effort nor miss the most effective training routine.
To achieve this, you need only two things:
- A reliable marker of biological aging
- Real-time feedback about your training’s effect on that bioAge marker
Then you can trial-and-error your way to the optimal training routine (or any other health behavior).
Spoiler alert: neither p53 nor any other molecular marker can do that.
I explain the p53 issue in more detail in Explanatory Note 2, but you can skip it if you just want to cut through the chase.
Explanatory note 2
p53 won’t help you. For two reasons:
-
- It’s an anti-cancer, not a pro-aging protein
- It’s unsuitable for laypersons and for everyday use
P53 drives cells either to self-destruct or to become senescent (think geezer cells).
That’s a good thing when it happens to mutated cells that would otherwise turn into cancer.
But it’s a bad thing if there are too many geezer cells left to hang around. While they won’t proliferate (which spells death to cancer), they secrete molecules that turn their direct neighbors into geezers, too. And this is what prematurely ages the tissue or organ to which these cells belong.
To put it simply, too much p53 is undesirable for the whole organism, as is too little of it [4].
How to train for health, functionality, and independence
In a previous post I explained in detail
- how to turn yourself into a one-person clinical trial,
- why pulse wave velocity (PWV) is your biomarker of choice, and
- which tool to use to make the whole process as simple as stepping on a special bathroom scale every morning.
Just scroll down to the “Solution” section (if you want to skip the first part of the post).
While that post talked about diet interventions, the principle of piloting your diet habits toward healthy longevity applies 1:1 to exercise, too.
When it comes to exercise, you have four levers to individualize and subsequently test the efficiencies of your training routines.
They are easy to remember:
- F = frequency
- I = intensity
- T = time (duration)
- T = type of exercise
Researchers have studied (group-based) how each lever affects various biomarkers of fitness and health.
What interests us here is their effect on the one key biomarker of endothelial function and vascular ageing – PWV.
The figure below summarizes what these studies tell us in general.
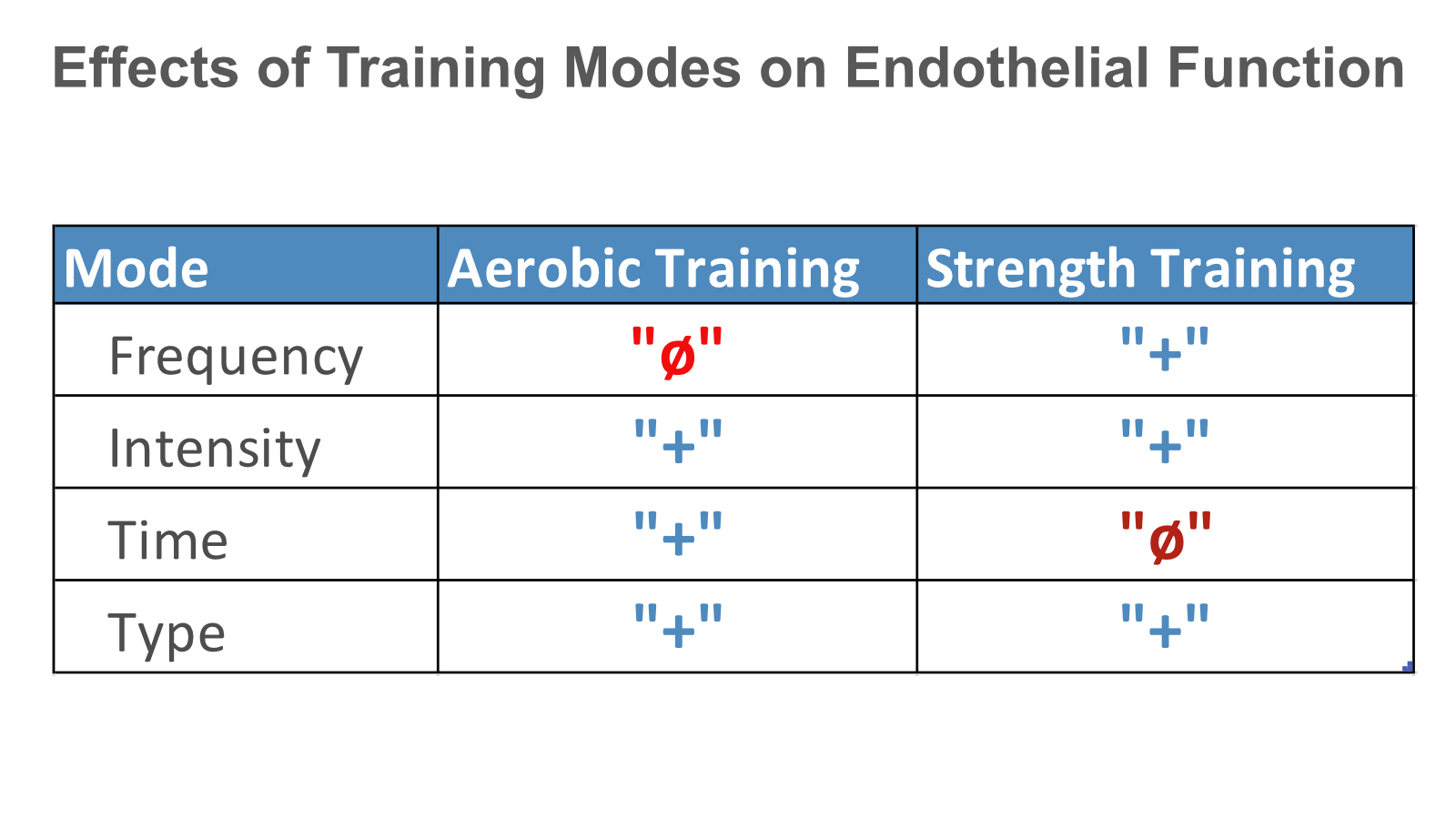
But as you know now, those results don’t necessarily translate to you.
This is why, in my next post, I will extract from all those studies:
- The range of possible effect sizes (so that you can prioritize)
- The profiles of the study participants (so that you can match them to yourself)
Both pieces of information will help you prioritize which levers to work on first. And you won’t have to rely on any health guru parroting study results that exceed their comprehension in the first place.
References
[1] Fisher AJ, Medaglia JD, Jeronimus BF. Lack of group-to-individual generalizability is a threat to human subjects research. Proc Natl Acad Sci U S A 2018;115:E6106–15. doi:10.1073/pnas.1711978115.
[2] Kent D, Hayward R. When averages hide individual differences in clinical trials. Am Sci 2007;95:60–8. doi:10.1511/2007.63.60.
[3] Rossman MJ, Kaplon RE, Hill SD, McNamara MN, Santos-Parker JR, Pierce GL, et al. Endothelial cell senescence with aging in healthy humans: prevention by habitual exercise and relation to vascular endothelial function. Am J Physiol Circ Physiol 2017;313:H890–5. doi:10.1152/ajpheart.00416.2017.
[4] Rodier F, Campisi J, Bhaumik D. Two faces of p53: Aging and tumor suppression. Nucleic Acids Res 2007;35:7475–84. doi:10.1093/nar/gkm744.